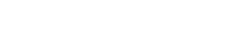A 5-day ex vivo pilot study using living human skin to generate early local toxicity and immune response data for your injectable therapeutic.
One donor. Clear safety signals. NAM-aligned.

Injection site reactions are the #1 reported adverse event for biologics
If you’re developing an injectable biologic, monoclonal antibody, ADC, or peptide therapeutic, injection site reactions are a foreseeable risk. They remain one of the most frequently reported adverse events in clinical trials for subcutaneous and intradermal products.
Standard pre-clinical models like murine skin lack the structural complexity, immune cell composition, and barrier architecture of human skin. Reactions that go unnoticed in animals can surface in first-in-human studies, leading to clinical holds, protocol amendments, or costly reformulation cycles.
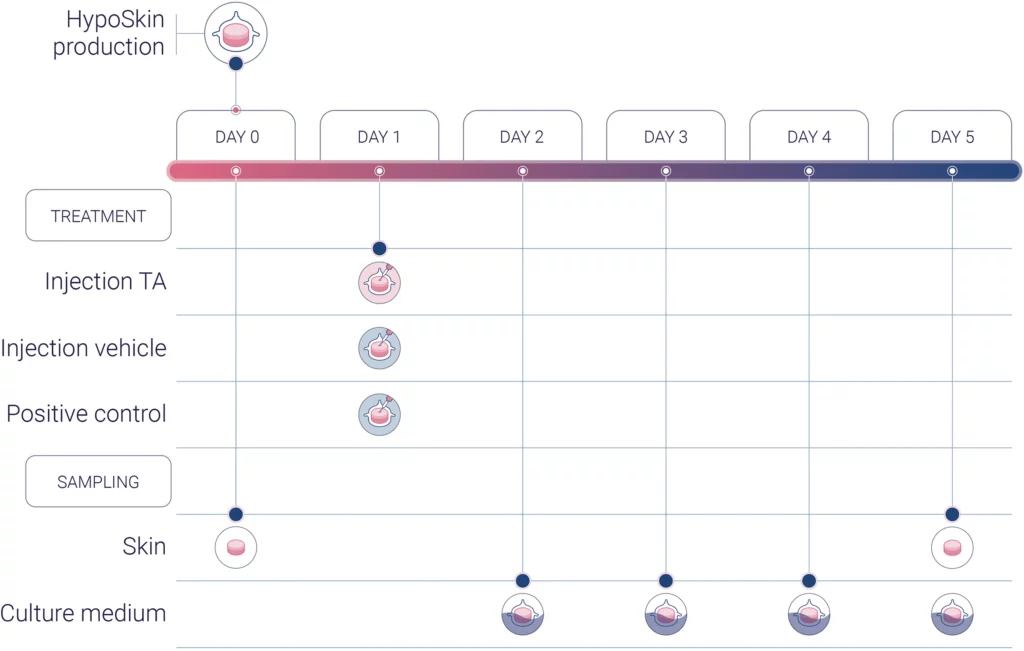
Experimental conditions
1 donor (n=3)
1 vehicle (negative control)
1 TA
1 positive control
Analysis
H&E staining
Multiplex cytokine analysis
Evaluate local toxicity using biostabilized human skin
Objective of the study
Evaluating local toxicity and injection site reaction in human skin is essential to de-risk injectable therapeutics before moving to clinical stages. Genoskin’s HypoSkin® platform provides an ex vivo, immunocompetent human skin model that preserves native structure, innate immune components, and subcutaneous architecture — delivering human-relevant insights that align with current NAM-focused regulatory expectations.
This toxicity pilot study offers an entry point to assess whether your therapeutic compound elicits morphological damage or initiates early inflammatory responses when injected into human subcutaneous tissue. Based on ex vivo biostabilized human skin, this New Approach Methodology provides decision-enabling data that support early safety assessment and guide downstream development.

This feasibility study is recommended when:
You are new to Genoskin’s technology and want to validate its relevance to your program.
You need early human-based data to support internal decision-making.
You require preliminary evidence to justify a larger, multi-donor investigation.
You are looking for preliminary data before setting up a larger program to de-risk several therapeutic candidates.
Analysis & readouts
READOUTS INCLUDED
Tissue viability: Hematoxylin & Eosin (H&E) staining
Immune response assessment: Multiplex cytokine analysis
SPECIFIC QUESTIONS ADDRESSED
Experimental workflow
Deliverables & important information
DATA PACKAGE
FINAL REPORT
Biological variability
Scope & limitations of use
- Any clinical and/or medical applications
- Attempts to discover composition, manufacturing processes, or trade secrets behind the materials
Ethical sourcing & compliance
Common questions about the pilot study
How is this different from testing in animal skin?
What is the typical turnaround time?
Can I draw reliable conclusions from a single-donor study?
How is the tissue sourced?
What types of injectables can be tested?
Does this data carry regulatory weight?
Ready to generate early safety data for your injectable?
Tell us about your compound and study objectives. Our scientific team will review your request and respond with a tailored pilot study plan including proposed timeline, readouts, and pricing, typically within 2 business days.
No commitment required. This is a conversation, not a contract.
Study plans designed by Genoskin’s in-house scientific team
Typical response time: 1–2 business days
Your information is confidential and protected